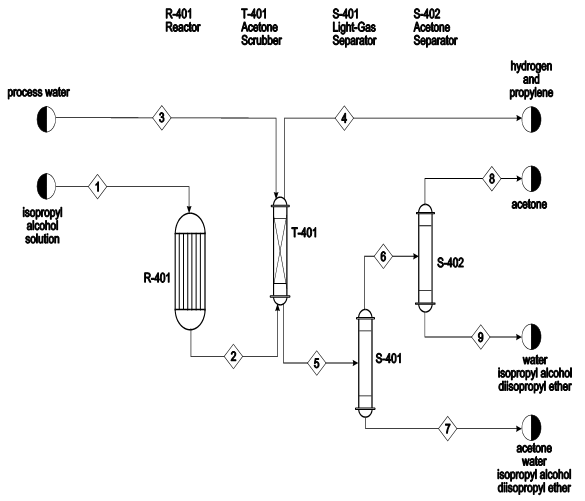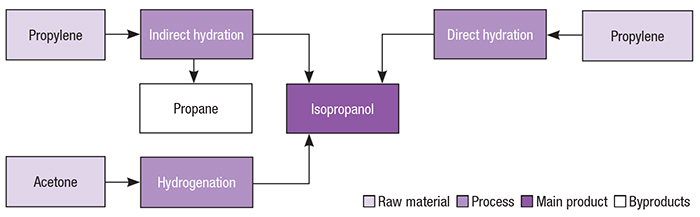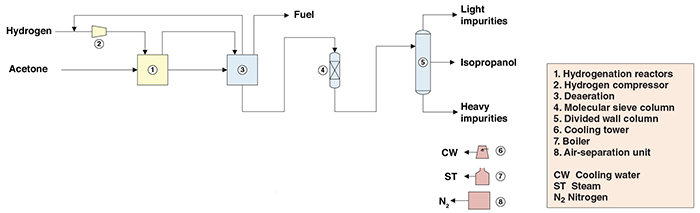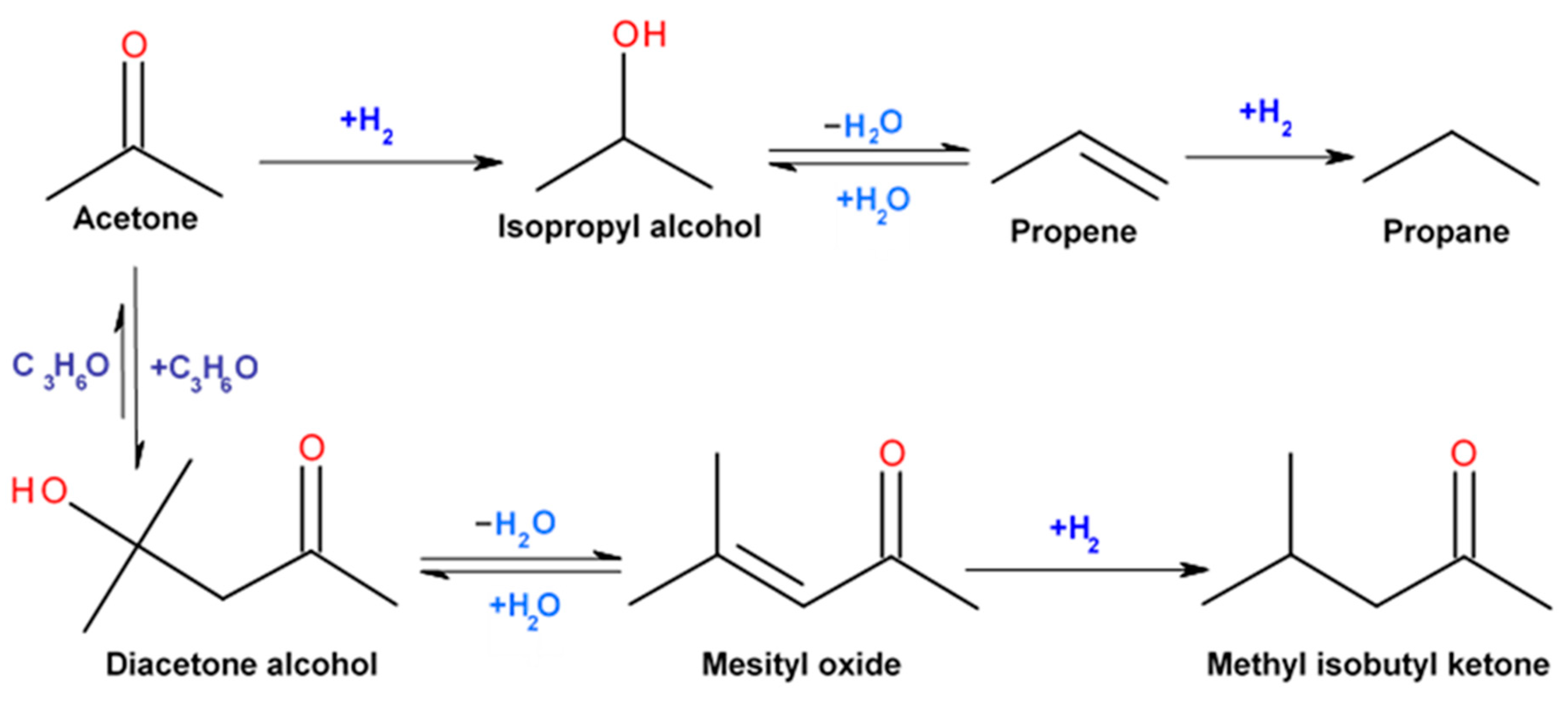
Catalysts | Free Full-Text | Highly Selective Gas-Phase Catalytic Hydrogenation of Acetone to Isopropyl Alcohol

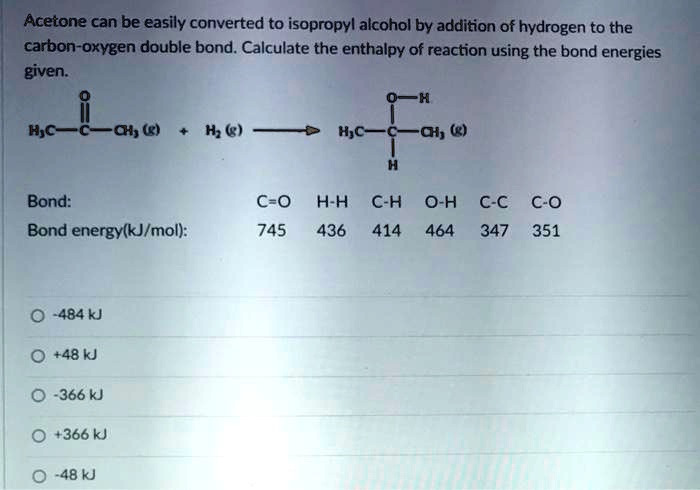
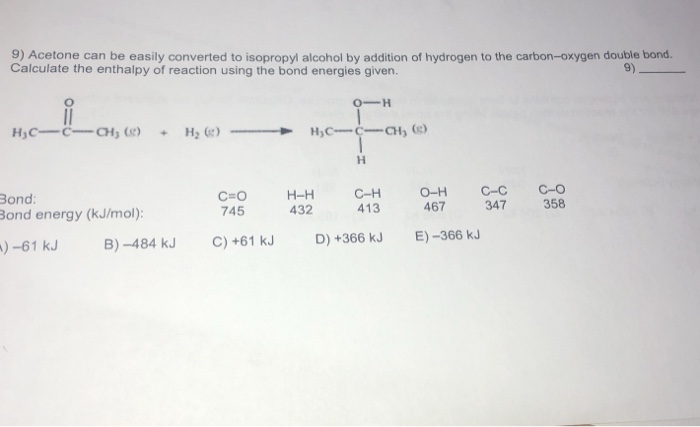
Acetone can be easily converted to isopropyl alcohol by addition of hydrogen to the carbon–oxygen double - Brainly.com
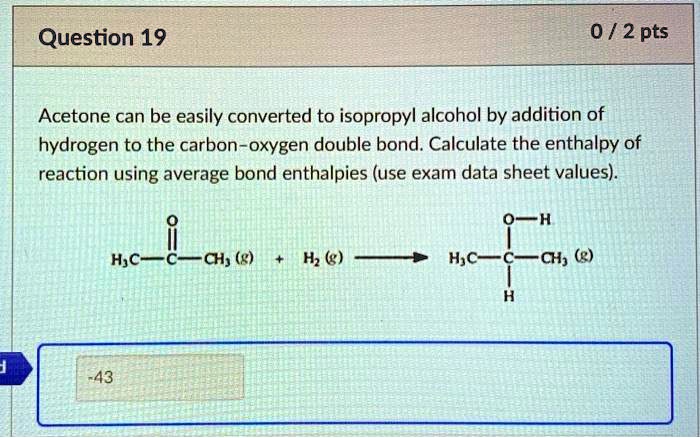
SOLVED: Acetone can be easily converted to isopropyl alcohol by the addition of hydrogen to the carbon-oxygen double bond. Calculate the enthalpy of the reaction using average bond enthalpies (use exam data